How much is a drug worth? The healthcare industry as a whole has not developed a consistent method for pricing drugs, though each side of the transaction has its own approaches and priorities on placing a value.
Payers want to pay as little as possible, and only for drugs that have been proven effective. Pharmaceutical companies want to justify whatever price they have attached to a drug, and may cite its cost of development, or compare the drug’s regimen to the cost of alternative treatments. Clinicians and patients rarely even know the price and tend to focus on three things when it comes to a drug: Does it work, how bad are the side effects, and is it covered by insurance?
If we don’t know how to measure the value and pay for a drug today, how can we hope to get prices under control for the next generation of therapies? The more we discover about the nature of disease and the more we use the vast datasets available with genomics, microbiomics, and metabolomics, the more the value question will dog us. These studies will inevitably encourage the development of new treatments and even cures benefitting ever smaller pools of patients; some may even be tailored to individual genetic profiles.
Hard to pay for
Six-and even seven-figure price tags are also inevitable because we are taking on more and more impossible-to-treat diseases. For example, the cancer immunotherapy drug Keytruda™ from Merck costs $150,000 per year, but offers hope to many who once would not have survived a year with their cancer. Recently, Novartis achieved landmark approval for the first CAR-T based cell therapy, Kymriah™. After demonstrating an unprecedented remission rate of 83 percent in a very tough to treat cancer patient population, it is expected to be priced at $475,000 per treatment.
If highly personalized drugs don't make it complicated enough to assign value, then what about conditions like HIV and many cancers that now need multiple drugs, often from different manufacturers? How do we compute the value of an individual agent in an HIV cocktail or a personalized breast cancer regimen? Should we even try? Initiatives like British Columbia's Personalized Onco-Genomics Program aim to create tightly tailored treatments. When each patient receives a different combination of drugs, a broad attribution of effectiveness becomes anywhere from impractical to impossible under current healthcare-financing models.
As payers and providers move to value-based reimbursement models, they cannot continue to operate without a consistent method of valuing drugs, because they will be unable to determine whether they should approve the use of a given drug for a given patient. Even if it's best for the patient, the payer may take a financial hit. Payers already face the problem of covering the cost of a cure that requires a significant upfront outlay (for example, Harvoni for Hepatitis C, with a price tag of $95,000), only to lose the now-healthy patient to another payer who reaps the entire benefit of that investment.
Under value-based care, we may inadvertently create incentives for under-utilization of some of our potentially most miraculous tools.
Sharing the risk
When providers start to share the payers' risk, they will get burned the same way. Physicians may hesitate to prescribe treatment regimens with front-loaded costs, depriving these regimens of the success that they may deserve for their overall clinical effectiveness. Under a fee-for-service system, we created incentives for over-utilization. Now, under value-based care, we may inadvertently create incentives for under-utilization of some of our potentially most miraculous tools, if we don't take precautions.
And what should those precautions be? Here are a few ideas:
Pay for the outcome, not the drug. If we agree on how much a disease should cost to treat by current methods, we can agree that a new drug costing less is a good value. In a partnership with the Centers for Medicare and Medicaid Services (CMS), Novartis agreed to absorb the cost of providing Kymriah™ to Medicaid patients if the patients do not respond within the first month of therapy. For diseases that are horrendously expensive to treat – late-stage cancer, life-long enzyme replacement therapy, or Hepatitis-C liver failure that requires a transplant as examples – the drugs could still be a good value even if their price is only slightly less horrendous than the treatments they replace. But the pharmaceutical companies involved would have to prove it.
Amortize the payments. If a drug maker wants five figures per treatment for a drug, the company should be willing to spread the cost over several years, during which the payer can track the patient's progress. If the patient switches insurance companies during the time of the payments, even if they have finished the treatment, the new payer would take up the payment schedule, thus ensuring that the original purchaser doesn't bear the entire risk. And if follow-up shows that the drug hasn't worked as promised, the maker would be required to forgo at least some of the remaining balance. This method would encourage drug makers to be rigorous in pre-market research regarding effectiveness and not oversell their products. It would also encourage better post-market research – a chronic Achilles' heel of our current system.
Be open to coalitions. Increasingly, treatments will have multiple components. Payers, providers, and pharmaceutical companies should work together and share the reward for good results. Under the current payment system, the pharma company is just another supplier, but as drugs become more personalized, their makers must function more like an active member of a care team.
The drugs now in development hold promises that we could not have dreamed of only a couple of decades ago. But we must find better ways to determine the value of those promises if we want them to be kept.
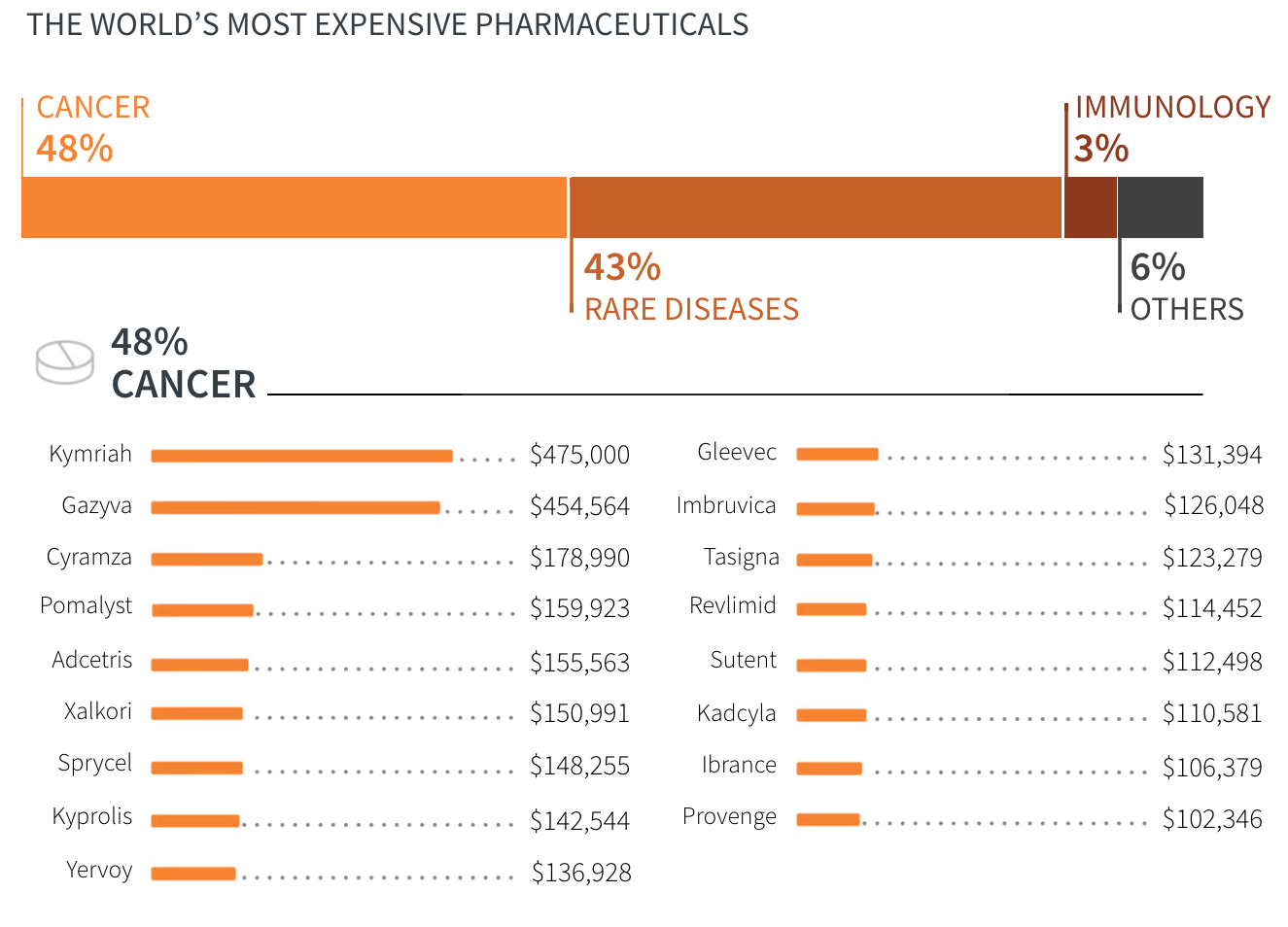
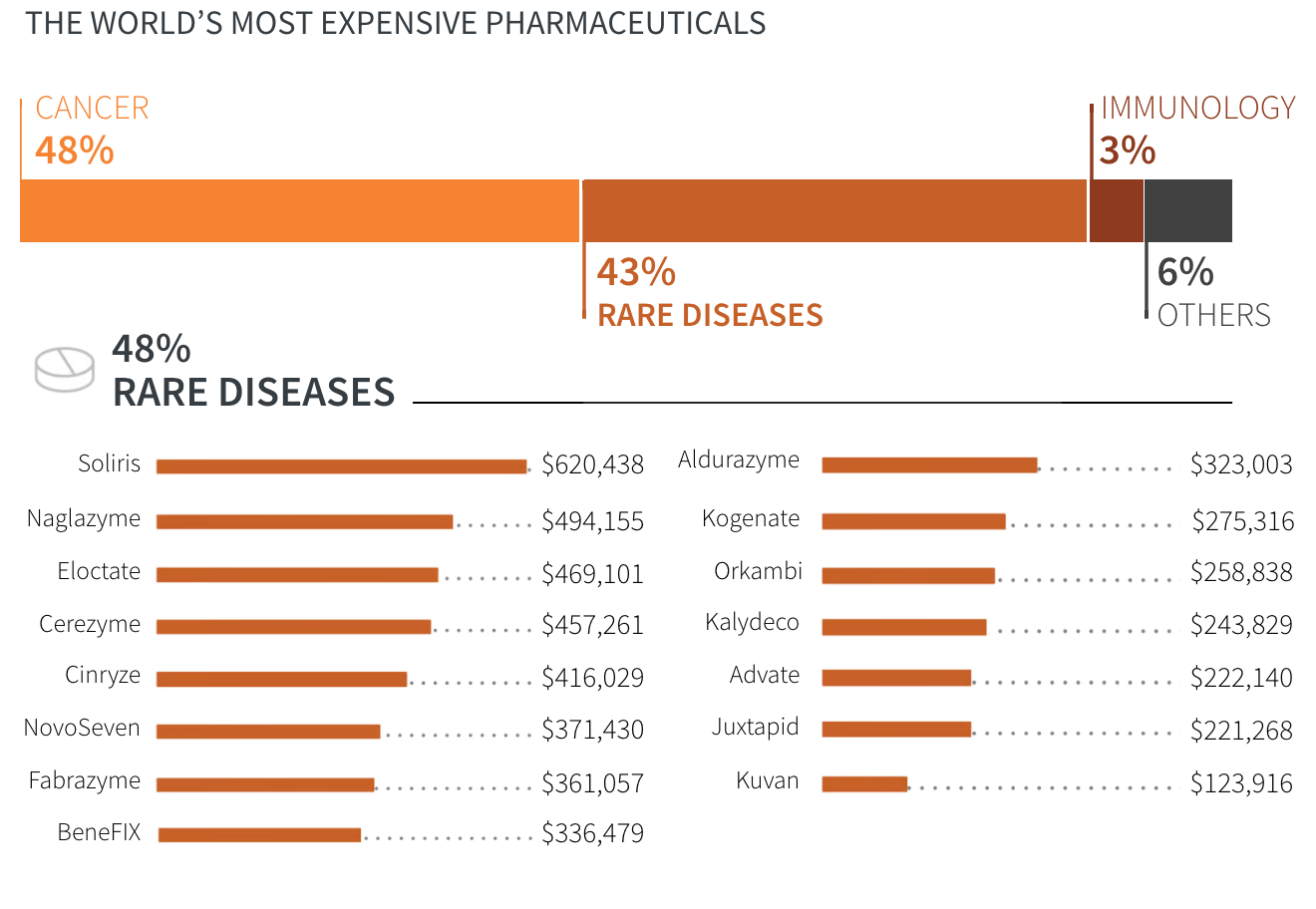
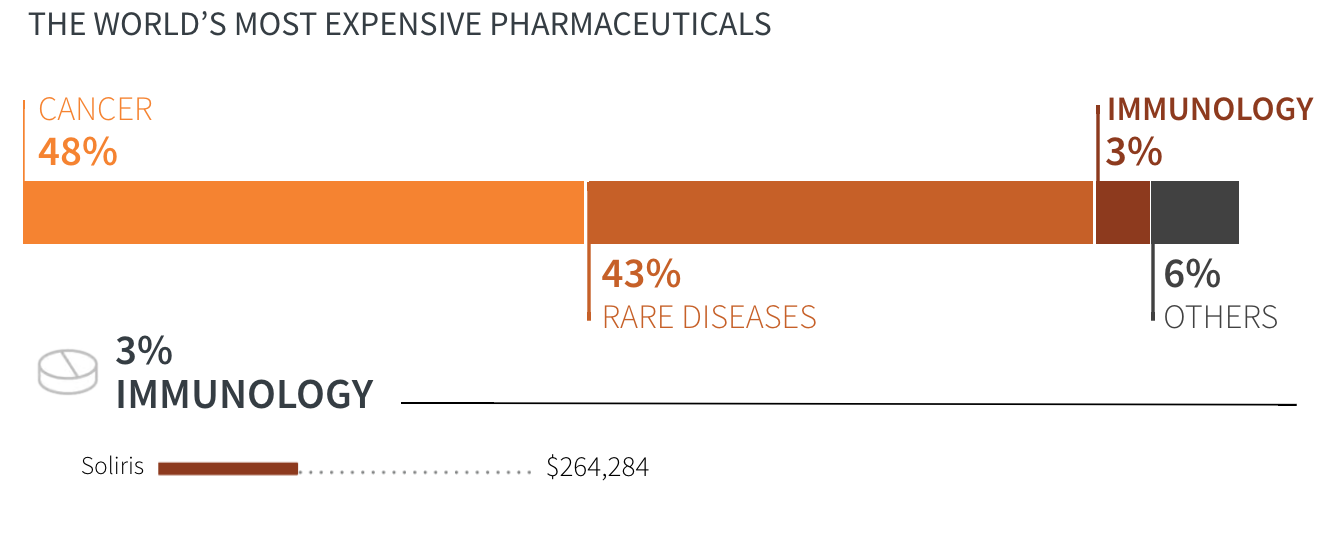
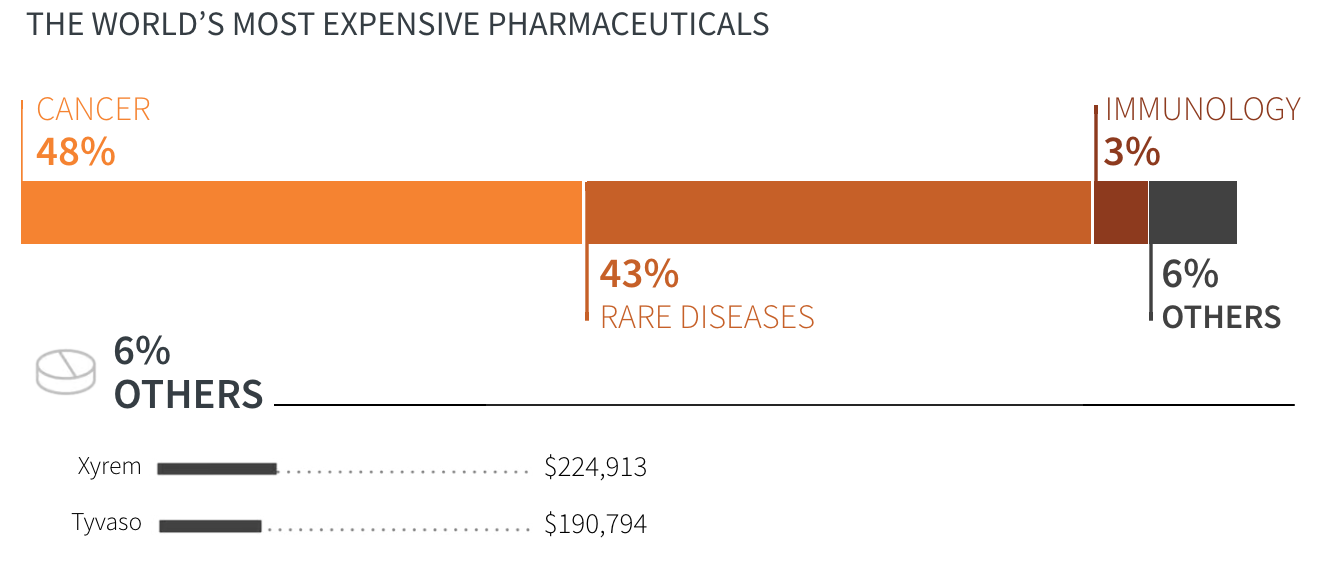
- Cancer
- Rare diseases
- Immunology
- Others
- Cancer
- Rare diseases
- Immunology
- Others
















